Understanding Cocrystallisation
Cocrystals represent a novel class of crystalline solids which consist of two or more molecular species usually held together by non-covalent bonds. Pharmaceutical cocrystal can alter the physicochemical properties of APIs to improve solubility, dissolution rate, particle characteristics and stability. Pharmaceutical co-crystals therefore possess scientific and regulatory advantages along with enormous intellectual property potential. Recently, the FDA approved the valsartan:sacubitril cocrystal Entresto as a dosage form in the treatment of heart failure. To obtain a successful pharmaceutical cocrystal product, it is crucial to understand cocrystal behaviour during manufacturing, storage, and administration as co-crystals can undergo different types of structural changes. The research team at CPES have many years’ experience in the study of cocrystals, investigating structural vs activity relationships and novel methods of manufacture.
CPES researchers have investigated novel manufacturing methods for cocrystallisation with significant cost saving and scale-up benefits over more conventional methods, for example:
- Continuous solvent free cocrystallisation via melt extrusion
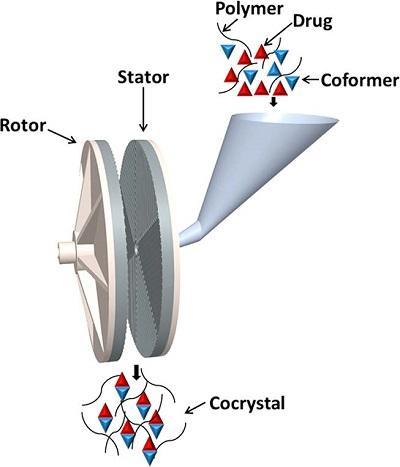
- Continuous cocrystallisation through solid state shear milling as shown in the figure opposite
- Microwave assisted cocrystallisation
- Ultrasound assisted cocrystallisation