Ran GTPase Inhibitors
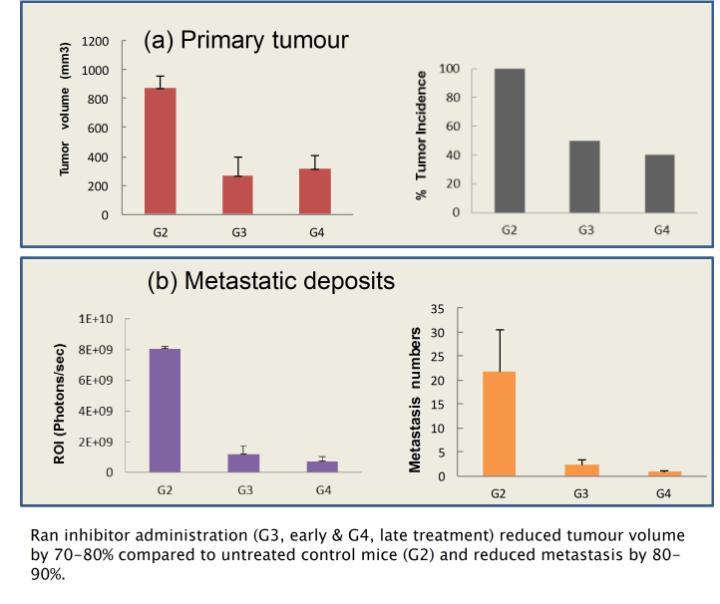
Ran GTP (Ran) is a Ras-related GTPase that is critical for mitosis, apoptosis and nucleo-cytoplasmic transport, and is overexpressed in breast and lung cell lines and tumours. High Ran expression in tumours has been shown to be associated with poor patient outcomes in breast, lung, ovarian and renal cell cancers. The project team have demonstrated that Ran expression can predict breast and lung patient survival and plays an important role in breast and lung cancer metastasis, highlighting a novel role for Ran in cancer progression.
The IP position
The patent for the use of RanGTP as a marker has been granted in most territories and a patent for its use as a therapeutic target in cancer has also been filed.
The opportunity
To greatly increase the potential value of the IP through studies to support its use as a target and/or marker in cancer. The next milestones is to progress a selected Ran inhibitor to first-in-man clinical trials.
For more information please contact Dr. Jason Jones or the UoB Commercial innovation Team